Solution to the carbon problem could be under the ground
Hope for the fight against climate change as study finds greenhouse gas can be buried without fear of leaking
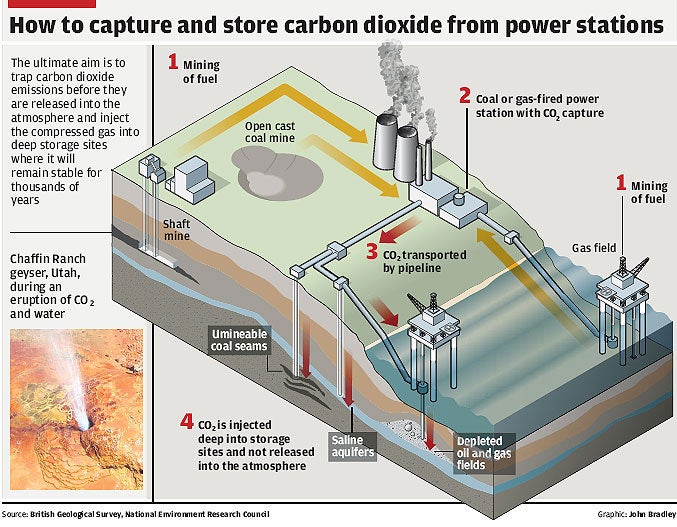
Carbon dioxide captured from the chimneys of power stations could be safely buried underground for thousands of years without the risk of the greenhouse gas seeping into the atmosphere, a study has found.
The findings will lend weight to the idea of carbon capture and sequestration (CSS) – when carbon dioxide is trapped and then buried – which is being seriously touted as a viable way of reducing man-made emissions of carbon dioxide while still continuing to burn fossil fuels such as oil and coal in power stations.
There are two substantial problems with CCS. The first is how to trap carbon dioxide efficiently in power-station emissions and the second is how to ensure that the underground store of the gas does not leak back into the atmosphere and so exacerbate the greenhouse effect and global warming.
In seeking to answer the second question, scientists looked at natural underground reservoirs of gas. They found that carbon dioxide trapped underground had been stable for possibly millions of years because it dissolves harmlessly in subterranean stores of water which do not appear to have leaked any substantial quantities of the gas back into the atmosphere.
The researchers believe the study shows that it will be possible to inject vast amounts of carbon dioxide from power stations into deep underground reservoirs where it will dissolve in water and remain undisturbed for at least as long as it will take for mankind to completely abandon fossil fuels and generate clean, carbon-neutral electricity.
Stuart Gilfillan of the University of Edinburgh said: "The study shows that naturally stored carbon dioxide has been safely stored for millions of years, which means that these sort of storage timescales should be achievable for the deliberate sequestration of the gas.
"It suggests that underground storage of carbon dioxide, in the correct place, should be a safe option to help us cope with emissions until we can develop cleaner sources of energy not based on fossil fuels," Dr Gilfillan said.
The study, published in the journal Nature, was based on an analysis of the chemical isotopes of helium and carbon dioxide in nine natural gas fields in North America, Europe and China. These gas fields have filled with carbon dioxide for many thousands or millions of years as it seeps from even deeper sources resulting from either volcanic activity or the heating of carbonate rocks.
The ratio of the two isotopes in the gas fields can tell the scientists whether any substantial quantities of carbon dioxide have seeped out of these underground sites during the period of time that they have filled up with gas.
Professor Chris Ballentine of Manchester University, who took part in the study, said that the isotopic technique will also be invaluable for further research, particularly when engineers begin carbon sequestration.
"The new approach will be essential for tracing where carbon dioxide captured from coal-fired power stations goes after we inject it underground – this is critical for future safety verification," Professor Ballentine said.
One of the reasons why the carbon dioxide remains trapped in the nine natural gas fields studied by the researchers could be down to physical changes occurring after its dissolution in water.
Dr Gilfillan said that when carbon dioxide dissolves in water the solution becomes denser than ordinary water and so sinks. This feature may have helped to keep the carbonated water underground for a long time, he said.
"We already know that oil and gas have been stored for millions of years and our study clearly shows that carbon dioxide has been stored naturally and safely in underground water in these fields," he said.
"It's good news in terms of the understanding of the system of carbon dioxide storage. It means that what actually happens in the natural storage of carbon dioxide suggests that it is possible to achieve the 10,000-year storage widely quoted as being necessary for effective carbon sequestration," he added.
There were initially fears that injecting carbon dioxide into the ground could simple result in it bubbling to the surface like a source of carbonated mineral water, releasing the gas into the atmosphere. The scientists also found that the underground carbon dioxide would not tend to form minerals and so form immovable solids. Mineral deposits block pores in rock, limiting the size of the overall carbon sink.
"It's bad news in the sense that mineralising the carbon dioxide would make it even more stable. But the good news is that mineralisation would have limited the amount of carbon dioxide that could be pumped into any one reservoir," Dr Gilfillan said.
Barbara Sherwood Lollar, a geologist at the University of Toronto, said that it was important to understand how carbon dioxide was stored in natural underground reservoirs if the problems of long-term storage of carbon dioxide were to be solved.
What we found was remarkable. At sites throughout the world, we found that the major way carbon dioxide is stored is by dissolution into the underground water, rather than by mineral trapping," Dr Sherwood Lollar said.
However, even if the sequestration part of the equation is solved, there is still the major problem of how to capture carbon dioxide emitted by power stations efficiently and cheaply.
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments
Bookmark popover
Removed from bookmarks