EU breast implant rules will create dilemma for No 10
Plan by Brussels to tighten controls would involve transfer of powers and could trigger referendum
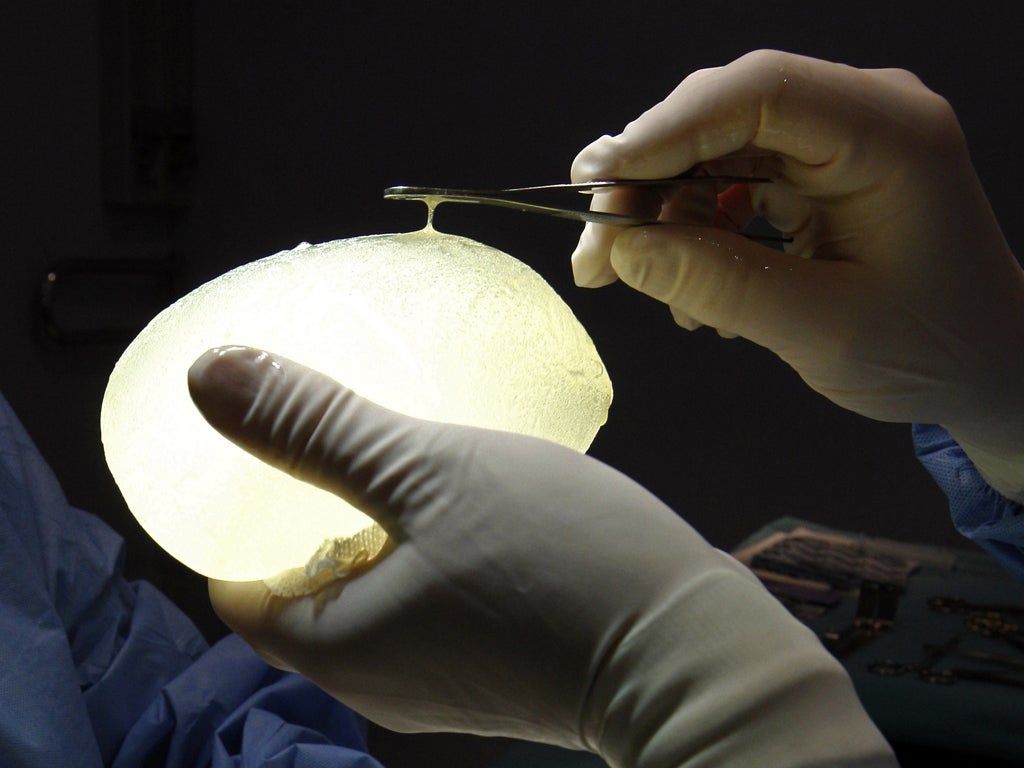
Senior figures in Brussels are drawing up new Europe-wide regulations to protect women against faulty breast implants in a move that could theoretically force David Cameron to hold a referendum, The Independent on Sunday has learnt.
Led by calls from Nicolas Sarkozy's government, a new EU directive on medical devices would tighten inspection regimes for silicone implant manufacturing companies and ensure each faulty implant could be traced back to production – two of the major factors behind the current French implants scandal.
But as the Government faced anger over its "loopholes" in the measures to help up to 40,000 British women affected, the Prime Minister was presented with a fresh dilemma over his tough stance on the power of Brussels to dictate UK national policy.
The European Commission has confirmed it is considering updating existing controls to ensure that every EU country applies the same tough measures to production and surgery.
The French health minister, Xavier Bertrand, said on Friday that tougher regulation "must be done at European level", adding: "Unlike drugs, where there is an authorisation on the market, there is not one today for medical devices."
A new EU directive would involve a transfer of national powers to Brussels, European experts said last night, which would technically mean the Prime Minister was obliged to hold a referendum on the issue in the UK. The Government has a get-out clause under which it could insist that the powers were not "significant" and would not need a national vote. But such a move, particularly one spearheaded by the French government, could provide fresh ammunition for Tory Eurosceptics.
The disclosure emerged after the Department of Health announced on Friday it would pay for the removal of implants for all 40,000 women who received the industrial-grade silicone devices manufactured by Poly Implant Prothèse (PIP). The Health Secretary, Andrew Lansley, said where implants were fitted on the NHS, it would pay for replacement, leaving taxpayers with a bill as high as £150m. But women who had implants fitted at private clinics would have only their removal funded – leaving potentially thousands of women forced to find more money if they wanted fresh implants.
Mr Lansley said private clinics had a "moral duty" to pay for patients who requested the operation. But Labour's health spokesman, Andy Burnham, said the Government's statement was "inconclusive". "By implication, the suggestion was that the best course of action is to have implants removed. But they provided no practical help to the vast majority of people affected. It is an unacceptable state of affairs for any woman now to be left in a position where she is worrying about her health, but is unable to afford to do anything about it."
Furthermore, the DoH has admitted it "cannot rule out the possibility that some are toxic". There were also reports of legal firms advertising for women to take part in class actions against the now-defunct PIP.
Denis MacShane, the Rotherham MP and former Labour Europe minister, said: "PIP was not adequately supervised by the French authorities and without a much tougher EU directive transposed into national law, we will see other scandals emerging, perhaps in new EU member states.
"Any new directive should also call for regulation of surgeons. In Rotherham, I am dealing with sad cases of women who had PIP implants and the private clinic won't see them for screening unless they pay more cash."
Officials from France and the UK are continuing to work together. "The UK would like to work with countries across the EU to understand fully where the failures of this incident fall," the DoH said.
The spotlight is expected to fall on the Medicines and Healthcare products Regulatory Agency over its handling of the crisis, in particular its reliance on dodgy data in the weeks after the PIP problems arose in France. Officials at the DoH are said to be frustrated at the slow speed with which the watchdog reacted.
The Commons Health Committee will meet Tuesday to consider launching an inquiry.
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments
Bookmark popover
Removed from bookmarks