Pioneering scientists grow rat's kidney in lab - and hope it will lead to breakthrough for human organ transplants
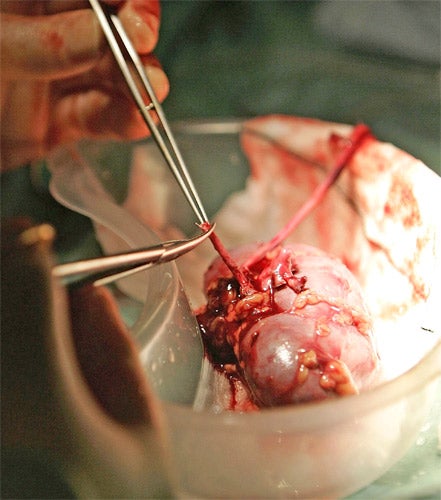
A synthetic kidney made from a patient’s own skin cells might soon be ready for the first human transplant following the creation of a similar artificial organ in laboratory animals, scientists said today.
Researchers created the synthetic kidney using a similar bioengineering process to the one that led to the manufacture of artificial human windpipes, the first of which was transplanted into a Spanish woman with a collapsed trachea in 2008.
The bioengineered rat kidney was made from the scaffold tissue of a dead rat’s kidney that had its own cells removed by flushing it with an enzymatic detergent. Skin and blood cells were then infused into the scaffold to create a working organ, scientists said.
Harald Ott of the Massachusetts General Hospital in Boston said that his colleagues have also created synthetic pig and human kidneys and that a human transplant of a bioengineered kidney made from a patient’s own cells is a realistic possibility. Such organs would not require immunosuppressing drugs because the tissue comes from the same patient receiving the transplant, Dr Ott said.
“What is unique about this approach is that the native organ’s architecture is preserved, so that the resulting graft can be transplanted just like a donor kidney and connected to the recipient’s vascular [blood] and urinary systems,” Dr Ott said.
“If this technology can be scaled to human-sized grafts, patients suffering from renal failure who are currently waiting for donor kidneys or who are not transplant candidates could theoretically receive new organs derived from their own cells,” he said.
Although there were a record 674 kidney transplants in the UK last year from donors who had died, and a further 1,009 transplants from living donors, there is still an acute shortage of kidneys. There are between two and four times as many patients awaiting transplants each year as there are donors.
Dr Ott and his colleagues created the kidneys by washing away the cells of organs taken from dead animals using a detergent that kept the organ’s connective tissue intact. He took cells from a rat foetus to make the blood vessels and the specialised filtration cells of the kidney.
A study in the journal Nature Medicine showed that the artificial organ was able to filter blood to produce urine, both externally and when transplanted inside a recipient rat. However, Dr Ott said that it did not function as well as a normal, healthy organ, possibly because the donor cells came from an immature animal.
“Further refinement of the cell types used for seeding and additional maturation in culture may allow us to achieve a more functional organ. Based on this initial proof of principle, we hope that bioengineered kidneys will some day be able to fully replace kidney function just as donor kidneys do,” Dr Ott said. “In an ideal world, such grafts could be produced on demand from a patient’s own cells, helping us overcome both the organ shortage and the need for chronic immunosuppression.”
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments