'Gene drive': Scientists sound alarm over supercharged GM organisms which could spread in the wild and cause environmental disasters
The development of so-called 'gene drive' technology promises to revolutionise medicine and agriculture

A powerful new technique for generating “supercharged” genetically modified organisms that can spread rapidly in the wild has caused alarm among scientists who fear that it may be misused, accidentally or deliberately, and cause a health emergency or environmental disaster.
The development of so-called “gene drive” technology promises to revolutionise medicine and agriculture because it can in theory stop the spread of mosquito-borne illnesses, such as malaria and yellow fever, as well as eliminate crop pests and invasive species such as rats and cane toads.
However, scientists at the forefront of the development believe that in the wrong hands gene-drive technology poses a serious threat to the environment and human health if accidentally or deliberately released from a laboratory without adequate safeguards. Some believe it could even be used as a terrorist bio-weapon directed against people or livestock because gene drives – which enable GM genes to spread rapidly like a viral infection within a population – will eventually be easy and cheap to generate.

“Just as gene drives can make mosquitoes unfit for hosting and spreading the malaria parasite, they could conceivably be designed with gene drives carrying cargo for delivering lethal bacterial toxins to humans,” said David Gurwitz, a geneticist at Tel Aviv University in Israel.
A group of senior geneticists have called for international safeguards to apply to researchers who want to develop gene drives, with strict security measures placed on laboratories to prevent the accidental escape of “supercharged” GM organisms that are able to spread rapidly in the wild.
Last week the US National Academy of Sciences initiated a wide-ranging review of gene-drive technology in “non-human organisms” and in this week’s journal Science a group of 27 leading geneticists call on the scientific community to be open and transparent about both the risks and benefits of gene drives.
“They have tremendous potential to address global problems in health, agriculture and conservation but their capacity to alter wild populations outside the laboratory demands caution,” the scientists say.
The researchers have drawn up a minimum set of safety rules to protect against laboratory escapes and have called for a public debate on the potential benefits as well as risks of a technology that allows geneticists to rapidly accelerate the inheritance of GM traits throughout an animal population within just a few generations.
Researchers have likened gene-drive technology to a nuclear chain reaction because it allows GM genes to be amplified within a breeding population of insects or other animals without any further intervention once the trait has been initially introduced. This is the case even if the trait is non-beneficial to the organism.
Laboratory experiments on fruit flies have shown that a modified gene introduced into one individual fly can take just a few generations to “infect” practically every other fly in the breeding population, in defiance of the normal rules of genetics which dictate a far slower spread.
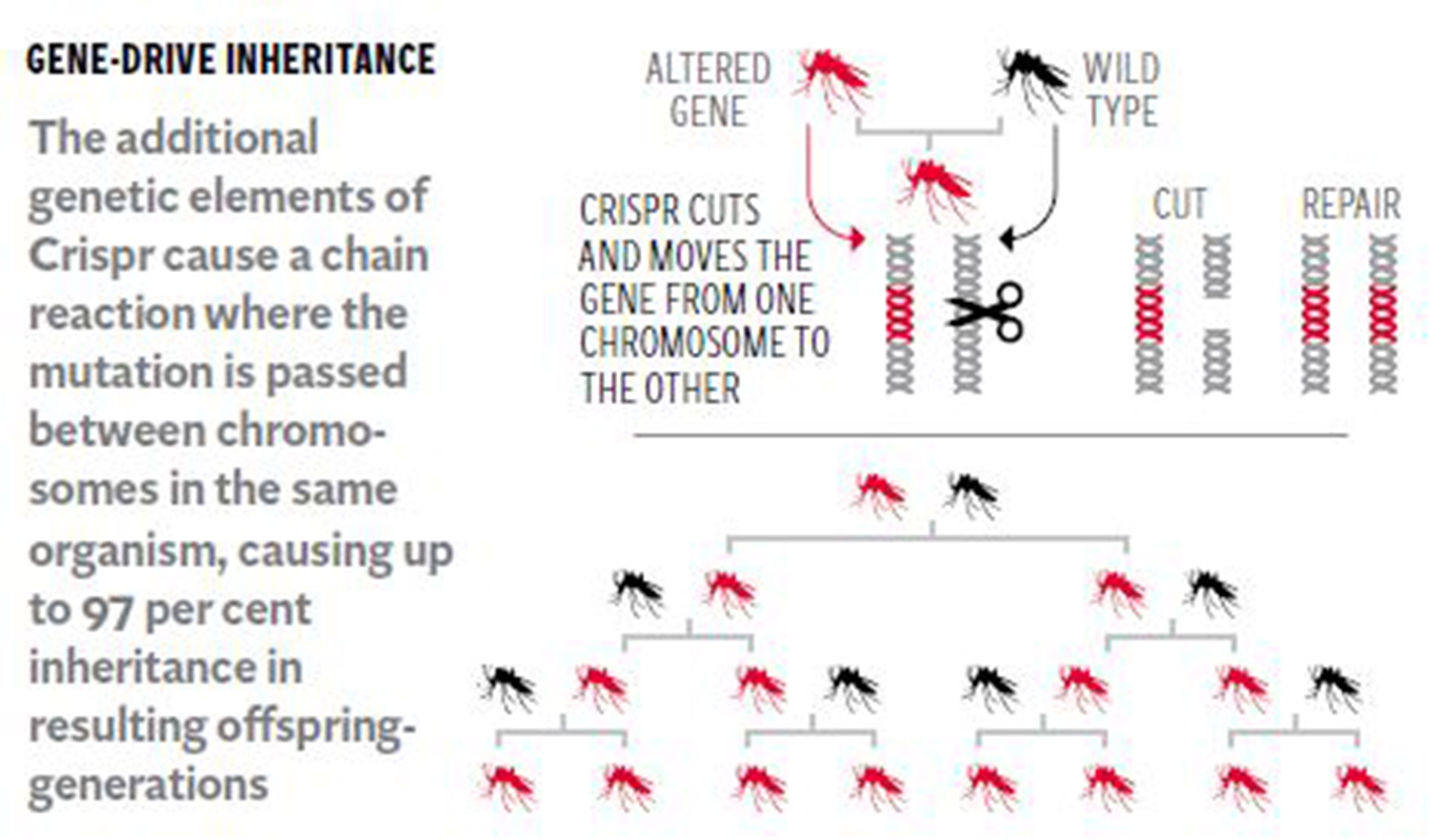
Kevin Esvelt, a gene-drive expert at the Wyss Institute at Harvard Medical School in Boston, said the technology was developed theoretically about 10 years ago but it has only been made possible in the lab in the past two years with the discovery of the sophisticated gene-editing tool Crispr/Cas9.
Dr Esvelt explained that gene drives relied on a “cassette” of genetic elements that allowed a genetically modified gene to jump from one chromosome to another within the same individual so that eventually all of the sperm or eggs of the animal carried the GM trait, rather than half. This means that virtually none of the offspring is eventually free of an introduced GM trait.
Gene drives could benefit human health by altering insect populations that spread human diseases, such as mosquitoes that transmit malaria, dengue, chikungunya and Lyme disease, so that they were no longer a threat, he said.

They could also be used to reverse the mutations that make crop pests resistant to agricultural pesticides, or they might be used to spread genetic traits within a populations of an invasive species to help kill it off, such as making the skin of cane toads introduced into Australia non-toxic to indigenous predators.
“If we’re right about this, it’s a powerful advance that could make the world a much better place, but only if we use it wisely,” Dr Esvelt said.
However, some scientists fear that the ease with which gene drives can be generated will make them a target for any malign individual or organisation with access to modern laboratory equipment.
Dr Gurwitz said the precise instructions for making gene drives should be classified, just like the technology for making nuclear weapons. However, Dr Esvelt and the other 26 scientists who have written to Science disagreed, arguing that complete openness and transparency was the best defence against the use of gene drives as a bio-weapon because classifying the information would be technically ineffective and politically counterproductive.
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments
Bookmark popover
Removed from bookmarks