Parkinson’s and cancer hope from implant drilled into skull to deliver drugs with pinpoint precision
‘It sounds as if she’s furiously sharpening a pencil inside my head’
A ground-breaking implant which allows drugs to be pumped into the brain could “massively expand” the treatment of neurological disease, strokes and cancer, scientists have said.
The valve system, which is screwed into the side of the skull, was developed by Bristol researchers as part of a landmark trial drug trial funded by Parkinson’s UK
The findings show “moderate to large” improvements in Parkinson’s disease patients, but little difference between those receiving the treatment, known as Glial Cell Line Derived Neurotrophic Factor (GDNF), and patients given a placebo.
This means the benefits may be down to the placebo effect or some other factor of the treatment, independent of the drug. And while there were some promising signs of nerve growth, the future of the drug is uncertain.
But independent researchers said that, regardless of GDNF’s results, the implant could have a “much wider impact” as the trial demonstrated it could be safely used to treat brain conditions in an entirely new way.
“This is a significant breakthrough in our ability to treat neurological conditions, such as Parkinson’s, because most drugs that might work cannot cross from the blood stream into the brain due to a natural protective barrier,” said the implant’s inventor Professor Steven Gill, from North Bristol NHS Trust.
Trials are already under way for a system delivering potent chemotherapy drugs directly in children and adults with incurable brain tumours, minimising the damage to other areas of the brain.
Once implanted, the only visible part of the system is a small port seated just behind the ear.
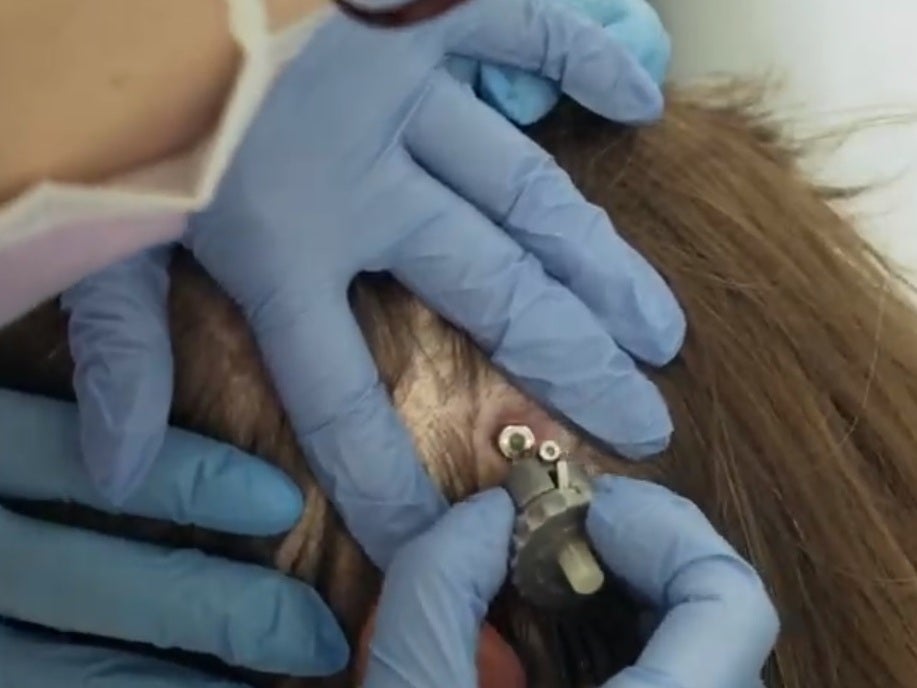
At each treatment session, the pump system is sterilised and screwed into the port, drugs are delivered via fine piping running over the surface of the brain to probes which route to the targeted parts.
“It sounds as if she’s furiously sharpening a pencil inside my head,” said Christine Proctor of the probe being attached, however once the drugs are being pumped there is no sensation of it in the brain.
Ms Proctor said the trial was one of the biggest risk she had ever taken, but she wanted to take part because there are currently no other options to halt or even slow the disease’s progress.

Researchers were hoping GDNF, a chemical produced naturally in the body which has been shown to help restore nerve damage, could regenerate neurons if pumped directly into the putamen.
The putamen is Brazil nut-sized structure in the midbrain affected in Parkinson’s and responsible for producing dopamine, the key neurotransmitter that is essential for coordinating movements as well as memory formation.
The death of these neurons in Parkinson’s causes gradual degeneration, which typically begins with the coordination of movement, facial expressions and posture, but it eventually affects other things like memory – causing dementia – and death.
Forty-one participants in the trial, published in the journals Brain and the Journal of Parkinson’s Disease, were randomly allocated to receive GDNF and or a placebo infusion. For nine months each participant had their movement, speech and other traits scored in Parkinson’s tests. The second half of the trial saw all patients receive the drug for a further nine months.
Over each year sufferers are expected to see around a one point decline, but patients receiving GDNF actually saw their scores improve by around four points in the first nine months, and nine points over all.
But disappointingly for those hoping GDNF is a cure, this was only marginally more than those receiving the placebo.
However, the Bristol team are not disheartened. Brain scans in the GDNF group showed some of the branching parts of the neurons that connect brain cells were regrowing – though whether this means the whole cell is healthy is less clear.

“We’ve shown with the Pet scans that having arrived, the drug then engages with its target, dopamine nerve endings and appears to help damaged cells regenerate or have a biological response,” said Dr Alan Whone, who led the trial at Southmeads Hospital.
“This represents some of the most compelling evidence yet that we may have a means to possibly reawaken and restore the dopamine brain cells that are gradually destroyed in Parkinson’s,” he added.
He said further trials were needed to understand why this regrowth was not replicated in symptoms and they hoped to test GDNF in higher doses – which they have now shown can be given safely – and over a longer period.
“The system developed for use here has been proven safe in practice over 80 weeks, which is a remarkable achievement,” said Professor Roger Morris, from King’s College London, who was not involved with the trial.
“If intra-brain perfusion can be safely achieved for long-term drug delivery inside the brain, the range of medication that can be used to combat brain diseases – stroke and cancer as well as neurodegeneration – will be massively expanded.”
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments
Bookmark popover
Removed from bookmarks