Infertile men 'could grow new sperm from skin cells'
Scientists produce healthy, fertile mouse offspring after creating sperm in laboratory dish with new technique
Infertile men could have their own biological children using a new technique that creates sperm from skin cells.
Scientists have created viable mouse sperm in a laboratory dish using the technique. The sperm cells were used to fertilise IVF mouse eggs to produce healthy, fertile offspring, which went on to have their own offspring by natural conception.
Experts believe it is the most convincing study yet to support the idea of using embryonic stem cells derived from a man’s skin cells to make his own sperm for fertility treatment. The sperm cells were derived from stem cells taken from a mouse embryo, but it may also be possible to use embryonic stem cells derived from adult skin cells, a technique that could allow infertile men to become fathers.
There have been several claims in the past of scientists creating sperm cells in the laboratory from adult or embryonic cells. However, this latest study is the first to conform to an agreed sequence of verifiable events to lead to the birth of healthy offspring.
“We established a robust, stepwise approach that recapitulates the formation of functional sperm-like cells in a dish,” said Jiahao Sha of Nanjing Medical University in China, where the research was carried out.
“Our method fully complies with the gold standards recently proposed by a consensus panel of reproductive biologists, so we think that it holds tremendous promise for treating male infertility,” Professor Sha said.
Making sperm cells in a test tube has proven difficult in the past because, like egg cells, they have to undergo a complex, two-step process of cell division called meiosis, where the 46 chromosomes of humans – or 40 chromosomes of mouse cells – are reduced to 23 chromosomes, or 20 in the mouse.
The Chinese team did this by recreating the natural environment of the body’s sperm-making machinery by co-culturing the cells derived from mouse embryonic stem cells with a cell suspension derived from the mouse testis.
Using this approach the researchers were able to convert cells called primordial germ cells into immature sperm cells, known as spermatids, which could be injected directly into an unfertilised egg to produce IVF embryos that went on to develop into healthy offspring.
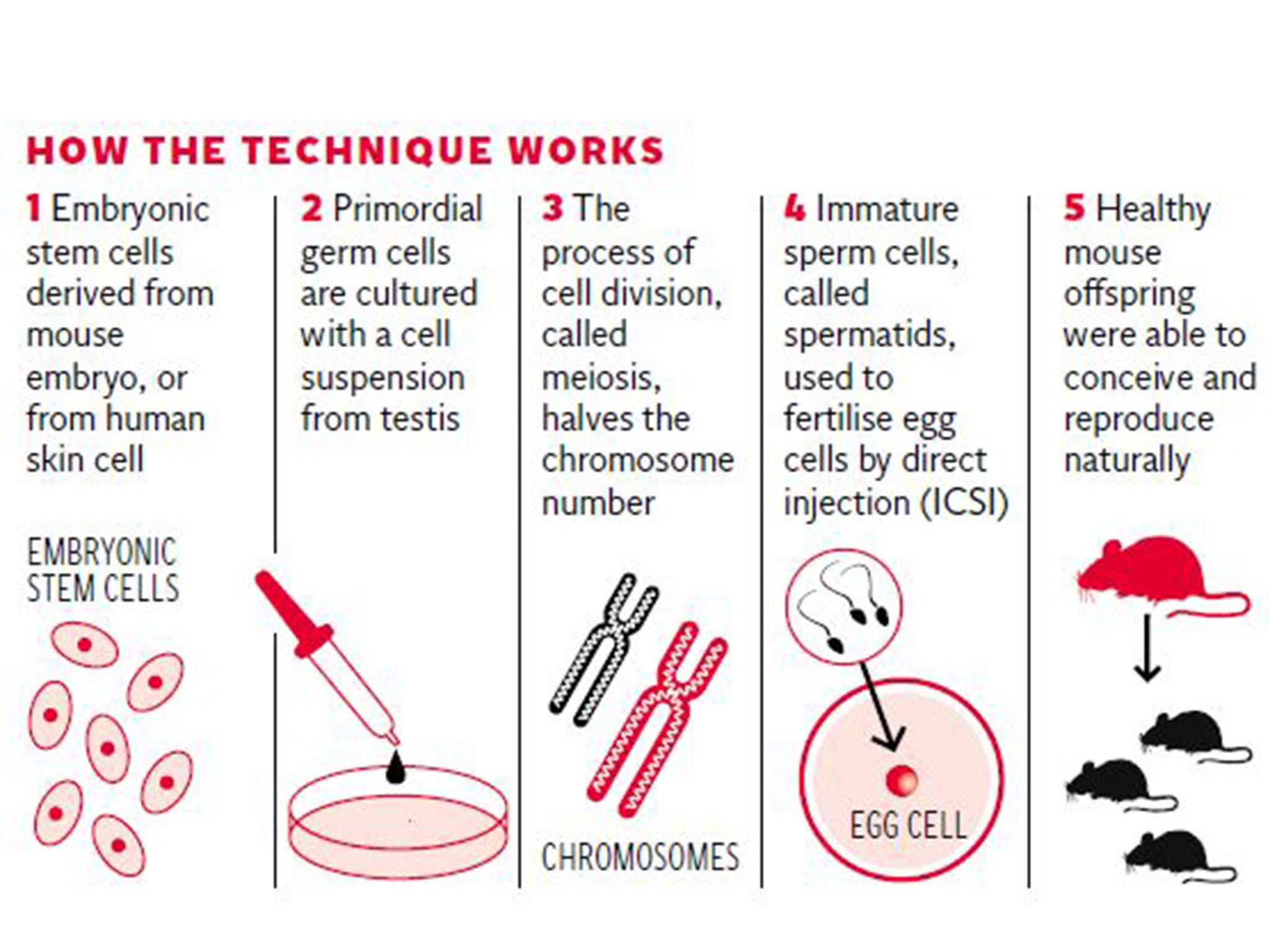
The study, published in the journal Cell Stem Cell, showed that these offspring were fertile and capable of conceiving naturally, the scientists said.
“If proven to be safe and effective in humans, our platform could generate fully functional sperm for artificial insemination or IVF techniques,” Professor Sha said.

“Because currently available treatments do not work for many couples, we hope that our approach could substantially improve success rates for male infertility,” he said.
Professor Simon Fishel, president of the Care Fertility Group in Nottingham, said: “This is exciting research that may represent a possible avenue in the longer term. However, establishing safety along with reproducibility will be the key before it can be considered as an option.”
Join our commenting forum
Join thought-provoking conversations, follow other Independent readers and see their replies
Comments
Bookmark popover
Removed from bookmarks